Data dashboard for supply decisions
The dashboard format keeps sustainability connected to purchasing behavior. A clinic cannot eliminate every single-use product, but it can choose better documentation, rotate stock correctly, reduce emergency shipments, and prefer products with clear packaging and take-back guidance.
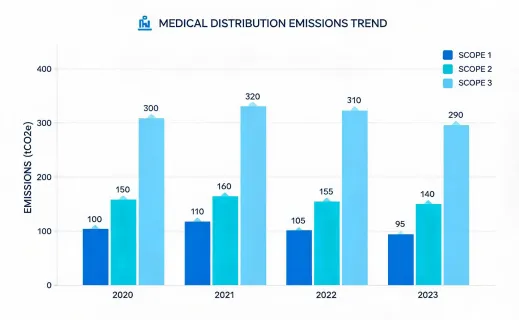

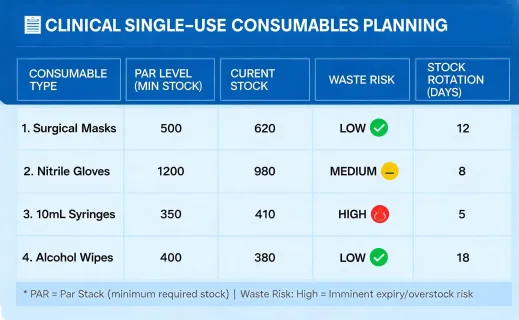
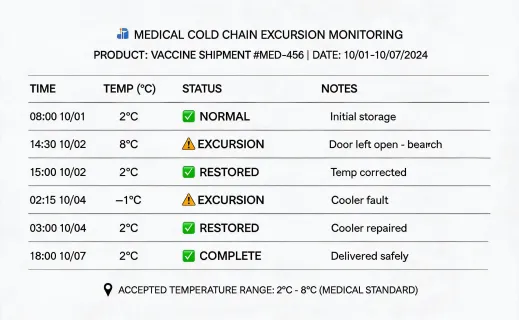
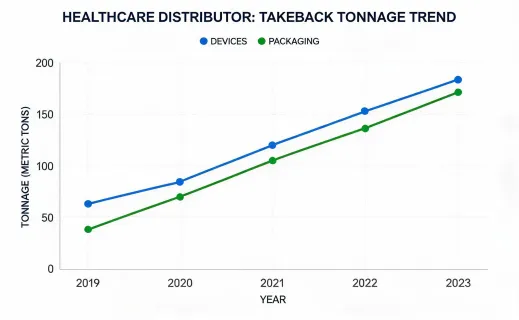
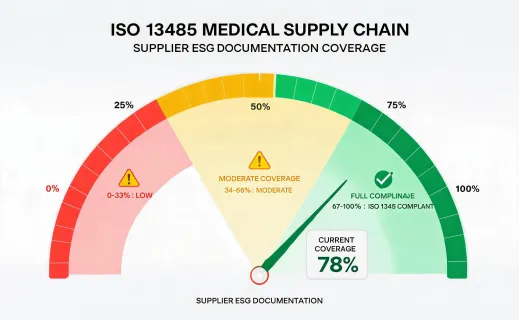
The sustainability program should not make unrealistic claims about sterile supplies. Many dental and infection-control products are single use for valid clinical reasons. The improvement opportunity is disciplined planning: fewer rush orders, better rotation, documented substitution rules, supplier packaging review, and accurate waste handling guidance. This approach gives procurement teams a defensible way to improve operations while respecting infection-control requirements.